Objective 1 : To Characterize the Skeletal Phenotype of Knockout Mouse Lines Produced by KOMP/IMPC
How different genes individually contribute to the architecture and composition of our skeleton remains a poorly understood area of biology; yet this information is fundamental to understanding the nature of complex skeletal diseases and provides the basis for therapeutic development. With the advent of the International Mouse Phenotyping Consortium (IMPC) and Knock Out Mouse Project (KOMP), initiatives that seek to disrupt the function of every protein-encoding gene in the mouse genome, we now have an opportunity to examine the skeletal phenotype of each genetically altered mouse line on an unprecedented scale. Given the high degree of genetic similarity between mice and humans, what we learn from the skeletal phenotyping of different mouse lines will have a significant impact on our understanding of the human skeleton. Thus, the objective of our team is to determine the bone phenotype of each knockout mouse line and report this information here. To learn more about the significance of this work, we encourage you to watch this presentation by Dr. David Rowe.
Objective 2 : To Establish a Repository Documenting the Bone Phenotypes of Different Mutant Mouse lines.
The scale of data being generated from large scale and individual bone phenotyping projects brings new possibilities to the manner in which we can understand and compare the impact that different genes have on the skeleton. Static and dynamic bone histomorphometry remains a broadly applied approach to phenotype the skeleton of mutant mouse lines. However, currently no central resource exists that contains bone histomorphometry data from different mouse mutants. Here we initiate an effort to establish such a resource here and invite members of the the research community to include the histomorphometry data they generated on their mutant mouse lines here.
Impact of Skeletal Diseases on Human Health and the Cost of Health Care
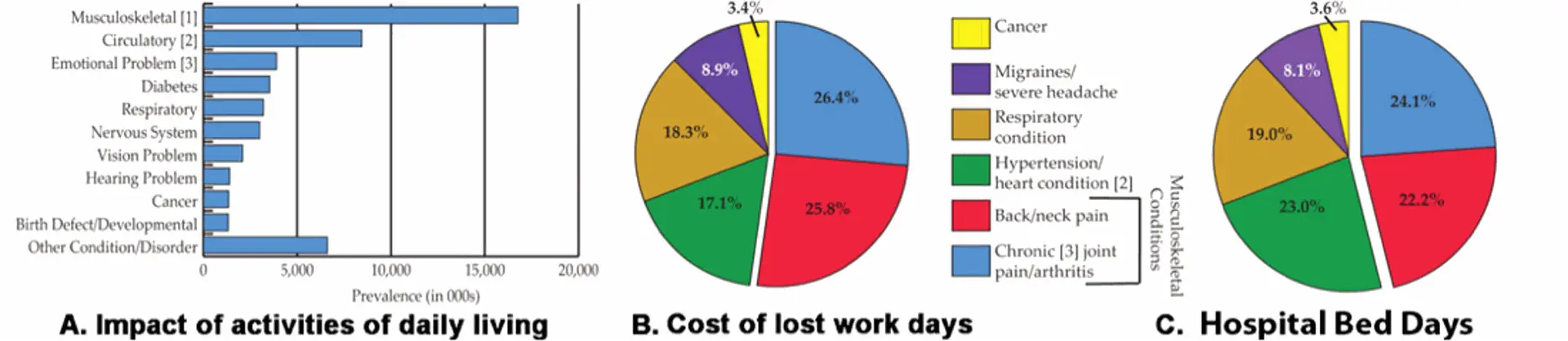
Heritable and degenerative diseases of the skeleton have a profound personal impact on the affected individual and place a huge financial burden on our health care system. Skeletal diseases and injuries are often extremely painful, limit physical mobility, and ultimately impact physical and mental well-being. While diseases affecting the skeleton are, in most instances, not life threatening, more than any other diagnostic disease category, they carry the highest burden to the health care system and to the economy overall due to their chronic disruptive impact on the quality of life (figure 1). As illustrated in the report, The Burden of Musculoskeletal Diseases in the United States carried out by the United States Bone and Joint Initiative, the cost of treating skeletal diseases represents approximately 7.4% of the country’s total GNP (45% x 17%). Because these non-curative treatments only assist the affected individual to cope with their disability, these conditions impose a continuous lifelong economic and personal burden. If our national goal is to mitigate this economic burden, we must identify individuals at risk for a skeletal disease before it develops. Once identified, the individual can either modify his physical behavior so as not to overly strain the vulnerable tissue, or institute a behavioral change or pharmaceutical intervention that helps the tissue compensate for its vulnerability. An example of this principle is the study of bone mass in twins that shows the importance of genetically determined accumulated bone mass prior to the onset of bone loss as a predictor of osteoporosis in elderly subjects. Since accumulated bone mass is acquired during the adolescent stage of growth, osteoporosis should be regarded as a pediatric disease. It follows that identifying individuals with genetic predisposition for suboptimal bone accumulation needs in the early childhood years so steps can be taken to enhance bone mass throughout childhood and adolescence. Thus the challenge for the basic research community it to provide the genetic markers for the primary care giver that are predictive of an adult disease outcome.
Overview of Skeletal Phenotyping Projects
Project 1: Bone Phenotyping of 220 Randomly Selected Knockout Mice
Project Period 2013- 2018: This project involved the skeletal phenotyping of 220 viable knockout mouse lines obtained from the knockout mouse project (KOMP). These mice were randomly selected based largely on their availability from the Jackson Laboratories production site for KOMP. For this project, animals were bred, injected with mineralization dyes, and euthanized at Jackson laboratories, then animals were sent to us for phenotyping. Knockout mice were phenotyped at 12 weeks of age (n=8 females, n=8 males). Initial screening for skeletal phenotypes was performed by micro-CT and if this analysis revealed a significant variation in bone parameters, histomorphometry was performed.
Project 2: Bone Phenotyping of Heterozygous Knockout Mice that as Homozygous Knockouts are Embryonic Lethal
Project Period 2019-Present: This is an active project involving the adult skeletal phenotyping of heterozygous KOMP generated knockout mice that in the homozygous state are either embryonic lethal and postnatally subviable. Mice from this project are selected based on a variety of criteria including gene expression, novelty, and/or some indications from initial IMPC phenotyping that a skeletal phenotype may exist. For this project, animals are being maintained at UConn Health in Farmington, so if you are interested in a particular KO line that is being actively maintained in our facility please contact us. Heterozygous knockout mice for this project will be phenotyped at 12 weeks of age (n=8 females, n=8 males). Initial screening for skeletal phenotypes was performed by micro-CT and if this analysis revealed a significant variation in bone parameters, histomorphometry was performed. We will also be performing a limited skeletal phenotyping analysis on late-stage homozygous knockout embryos.